Sudan and Chad: MSF treats 116 wounded in one month and warns of increasing casualties
Drone strikes near the Sudan–Chad border drive surge in civilian casualties
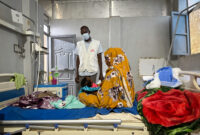
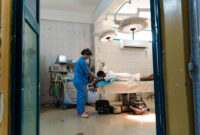
Drone strikes around the Sudanese town of Tina, have intensified since early May, leading to influxes of wounded patients across the border in Chad at the nearby Tiné hospital, which is supported by Doctors Without Borders/Médecins Sans Frontières (MSF). In recent weeks, residents have reported near-daily strikes, attributed to Sudan’s Rapid Support Forces, with as many as five or six strikes occurring on some days.
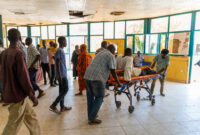
Since the beginning of May, 116 people wounded in these strikes were treated at Tiné hospital. Between May 17–26, 69 wounded patients were admitted. On May 24, a strike hit a busy cafeteria in Tina market, resulting in the arrival of 35 wounded people in a single day. Three people were declared dead on arrival at the hospital, while several others reportedly died at the scene. Women and children were among the casualties.
“In recent days, we have seen more and more women and children among the wounded.”
Issiaka Abdou, MSF country director in Chad
“The injuries we are treating are often extremely severe, including serious burns, blast-related trauma and multiple injuries,” says Issiaka Abdou, MSF country director in Chad. We recently treated a child with severe burns to the face, arms and legs. Every delay in evacuation reduces the chances of survival for the most critically injured patients.”
MSF medical teams are also observing a worrying shift in the profile of patients being treated.
“In recent days, we have seen more and more women and children among the wounded,” says Abdou. “On May 26, all patients received following drone strikes were civilians. This shows just how exposed people living in this border area are to violence.”
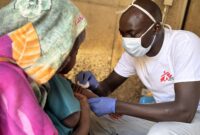
“Health facilities in this region are operating in an extremely challenging environment, with high needs and limited resources. Despite these constraints, our teams continue to provide emergency medical care and respond to influxes of wounded patients in coordination with the Chadian health authorities.”
Cissé Boucari Hamadoum, MSF project coordinator in Tiné
Rising needs overwhelm health services in Chad–Sudan border region
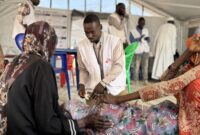
The border region between Chad and Sudan is severely affected by the conflict in Darfur, displacement of people and limited access to essential services, including healthcare. Repeated strikes are further exacerbating an already critical humanitarian situation.
“Health facilities in this region are operating in an extremely challenging environment, with high needs and limited resources,” says Cissé Boucari Hamadoum, MSF project coordinator in Tiné. “Despite these constraints, our teams continue to provide emergency medical care and respond to influxes of wounded patients in coordination with the Chadian health authorities.”
MSF expands emergency care
MSF provides medical care to patients at Tiné hospital and, when necessary, organizes referrals to other medical facilities, including in Abéché, for the most severe cases.
Civilians should never pay the price of conflict. It is essential communities are protected and wounded people can access medical care quickly and safely. Since the start of the war in Sudan in April 2023, more than 900,000 Sudanese refugees have sought refuge in eastern Chad. In response to this crisis, MSF has expanded activities in the provinces of Sila, Wadi Fira and Ouaddaï.
In eastern Chad, MSF teams are working in Adré, Ouré Cassoni, Iridimi, Touloum and Aboutengué, as well as in the surrounding villages in Ouaddaï province and in Tiné in Wadi Fira province, providing emergency medical care and supporting health facilities that serve both refugee and host communities.
Related news

Sudan: No safe place for women and girls in Darfur, MSF report finds